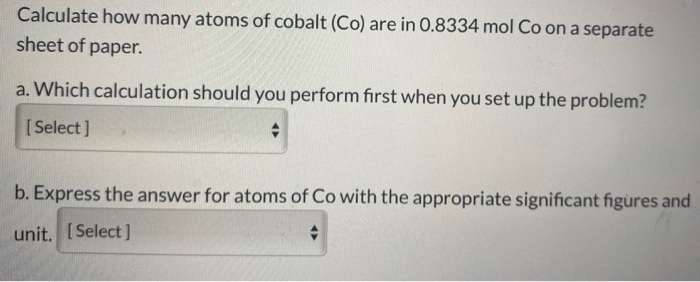
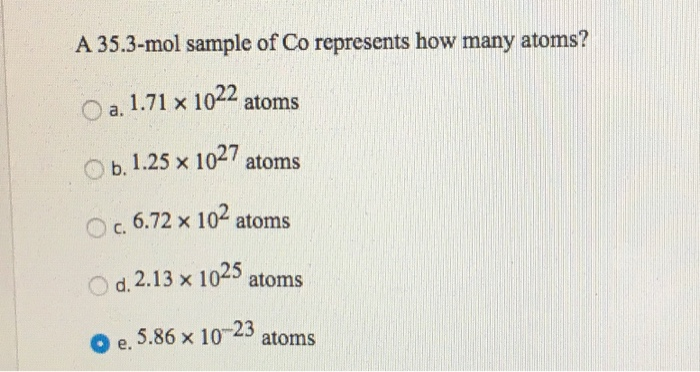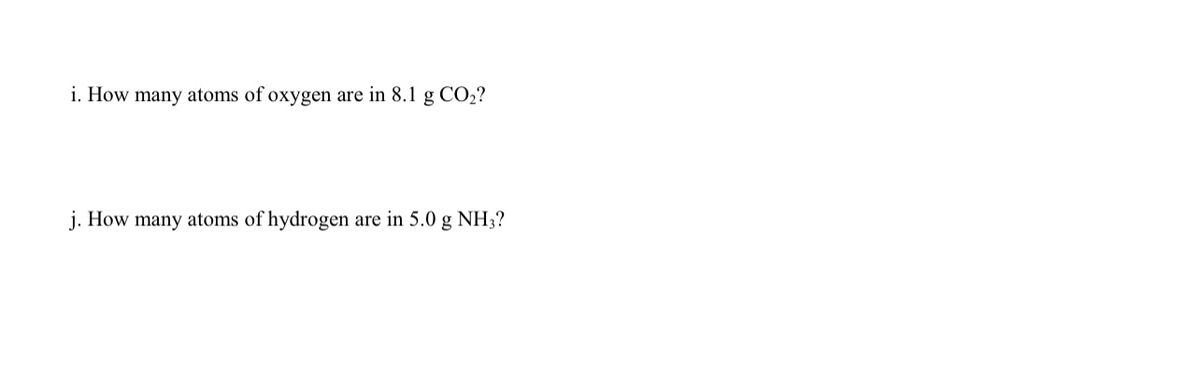QUESTION: How many atoms of oxygen are in 1.50 mol CO2? A. 1.50 mol, B. 0.300 mol, C. 3.00 mol, D. 0.750 mol 1 molecule CO2 is made up of 1 C

How many atoms are in 3.6 mol of calcium? How many moles are in 1.45 x atoms of sodium? What is the molar mass of K 2 SO 4 ? How many grams. - ppt download

Problem 3.14 How many moles of cobalt (Co) atoms are there in 6.00 X109 (6 billion) Co atoms? - ppt video online download